CRO Services By your side, every step of the way
LAHAV C.R.O (contract research organization) is Israel’s largest cro pre-clinical center specialized in large animal studies. Your one-stop shop throughout the pre-clinical phase, we flexibly provide full support or customized programs from proof-of-concept to large-scale GLP.
PLANING
Define the project with our experts
Consult with our pre-clinical C.R.O experts to make the right research decisions, right from the start.
Learn about the latest methodologies, new tools, and established best practices. Together with our professional team, we can quickly structure the study, review the protocol, and then choose the ideal animal model.
PREPARATION
Valuable guidance and proven methodologies prepare you for the study.
After facilitating decades of various studies with uniquely demanding challenges, we’ve developed a knowing knack for assisting clients to avoid missteps and mistakes.
Our professional preparation includes:
- Defining the work.
- Ethical submission and approval.
- Writing the protocol.
- Preparing animal follow-up documents.
Create your checklist we’ll get things done


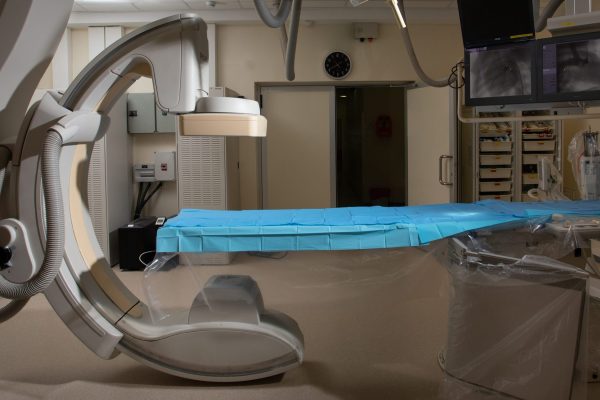

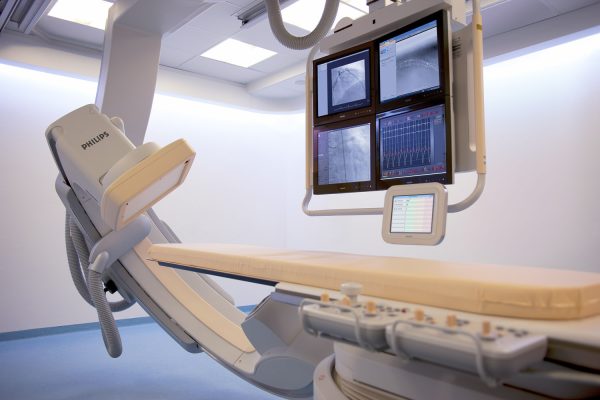

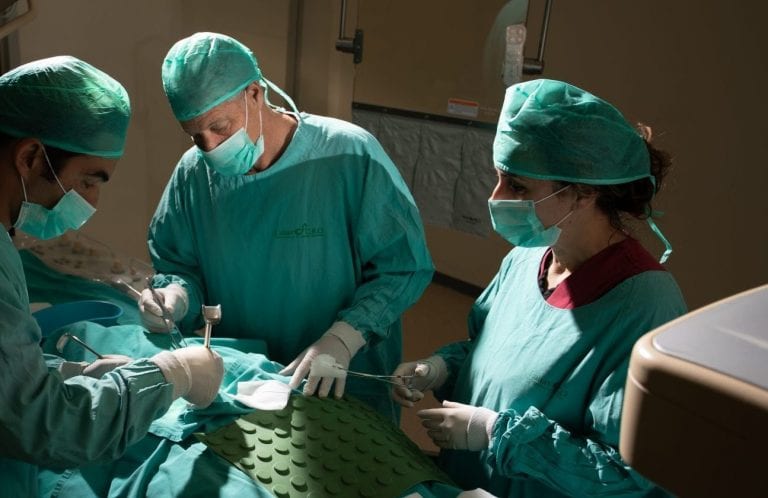

EXECUTION
Enjoy our full support on
the day of the study
LAHAV’s fully equipped operation rooms and medical device cro offer complete surgical suites and advanced imaging.
Exceptional surgical staff and veterinarians support you before, during and after the study.
We are insightful professionals who can quickly resolve unplanned issues and last-minute surprises.
FOLLOW-UP
24/7 vet care
- Twice-daily animal technician inspection by our professional staff
- Immediate and/or fast on-call veterinarian care around-the-clock.
- Real-time reporting on the research subjects for true peace-of-mind.
- Archival animal data reports document everything for your private reference.


CONCLUSIONS
Conclude your research with organized results
What are you interested in?
Our Therapeutic Areas
More Therapeutic Areas
Testimonials
Our clients stories










Contact Us


Publications

Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis
Ramot, Y. Dolkart, O. Steiner, M. Jahn, S. Goldberg, R. Cacical, O. Lavie, Y. Ezov,
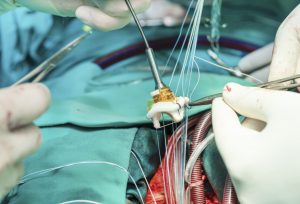

A Translational ‘Humanised’ Porcine Model for Trans-Septal Mitral Valve Interventions: the Right Pulmonary Vein Approach
Avner. A. Sabbag, I. Butnaru, A. Meerkin, D. A Translational ‘Humanised’ Porcine Model for Trans-Septal


Saliva metabolome alterations after acute stress
Morgan, L. Isak, R. Birkler, D. Shaham-Niv, S. Dong, Y. Wachsman, T and others. Saliva
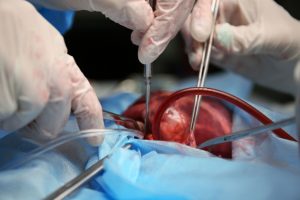

a Novel Transcatheter Ventricular Repair Device
W.Stone, G. Sievert, H. Virmani, R. Waisman Shelar, L. Manash, B. Neustadter, D. Description, Feasibility,
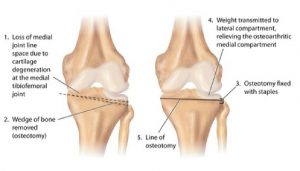

Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis
Ramot, Y. Dolkart O. Steiner, M., preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for


A novel fully tapered, self-cutting tissue-level implant: non-inferiority study in minipigs
Chaar, EL, E. Puisys, A, Sabbag, I. Bellon, B. Georgantza, A, Kye, W. E, Pppenger,


ETHICS CODEX


Quality and Service
Good Laboratory Practice


Ethics







