Accelerating Your Preclinical Development Study– from early stage proof of concept to large-scale GLP. Our CRO (Contract Research Organization services include: Study design, ethical submission, model selection, protocol writing, drug and medical device performance evaluation, safety & efficacy. Professional accompaniment up to the stage of the submission of the final report.
Our experts committed to addressing all your needs, operating with high reliability and standardization, providing innovative tailored solutions, cost and time effective. We have structured a cutting-edge hospital-environment facility, a technology platform, allowing a successful translation of pre-clinical assets towards the clinical evaluation phase.
R&D studies – Preforming innovative activities and studies, supporting our customers to develop new products or improving existing products.
Facilities Use Only – We offer companies the opportunity to carry out their studies in our cutting-edge facilities, making use of our technology platforms and knowledge and at the same time enable them to conduct their studies by their own professional team, having close supervision of our veterinarians with absolute privacy. More and more companies are doing a rapid use of our facilities in order to train their doctors, technical staff, engineers and product developers.
Controlled studies – Our study directors will write the protocols, prepare test documentations, conduct the trials and final reports.
GLP- Good Laboratory Practice – We will conduct the trials, perform and ensure the tests with uniformity, reliability and reproducibility.
Other Complementary Services and Documentation:
General services:
While Medical Device segment become more evolving advanced technology space, we are offering a high diversity platform for the development process, performing efficacy and safety testing, safety monitoring and proof of concept. Having an extensive experience in the evaluation of performance & safety of medical devices with strong expertise in the cardiovascular space. Our expertise includes: Biocompatibility & Efficacy, open surgery, interventional procedures, diverse device implantations, imaging and pathology. Our added value of better technologies, cost savings and potential for minimizing risks.

With massive growth in the pharma industry: rise in the number of complex drugs entering preclinical trial, growing pressure to reduce R&D expenses and attrition of drugs due to toxicity, we are offering full package of pharmacovigilance cro services, including efficacy, pharmacology toxicology, and safety pharmacokinetic studies. Our end-to-end services are of better quality, established scientific experience, economic stability and better logistics. Our experienced team helps companies to accelerate their preclinical drug development by providing a wide array of safety and efficacy assessment services complemented by our cutting edge innovative translational disease animal models (pig) and surgical research capabilities. We also provide complimentary histology and clinical pathology services


Breeding and Supply of Pigs organs
Several species of pigs ( Swine, Hog, Grunter, Porcine) from mini-pigs (Sinclair mini- pig, Gottingen mini pig) to fully grown domestic pigs with high variation in size & type of pigs (12.000 pigs), all pigs stress free, health monitoring.
Sheep
Several species of sheep.
Supply of Ex-vivo Organs
We supply pigs, Organs and tissue.
we are a worldwide recognized leader in preclinical surgical research conducted on large animals with significant track record in both catheter-based, minimal invasive and open surgical approaches. Our work has contributed to the development of multiple innovative products that have reached the market.
We have strong expertise in the cardiovascular space and other areas including dermatology, gastrointestinal, metabolic disorders, renal & urinary, respiratory, dental medicine, orthopedic, pain management and drug delivery.
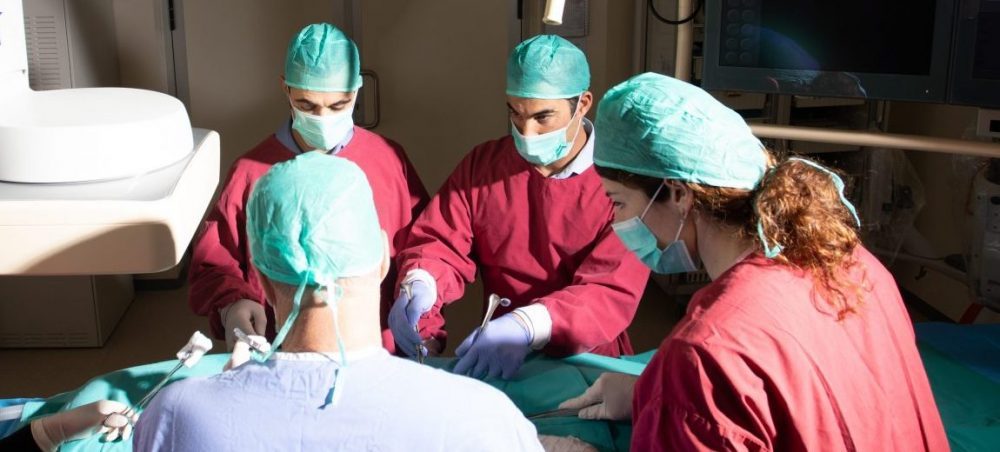

Physician Training:
We have a unique training platform and know-how, for doctors and first aid providers with strong expertise in emergency and trauma procedures. In the reality of living under daily trauma, we have witnessed testimonies from first aid providers and doctors, which had been trained in our facility and with our team, that these trainings, contributed to their ability to save people’s lives in real time.
We make sure to supply the appropriate level and access to medical and pre-clinical knowledge, expertise and ongoing collaborations with Doctors and academic institution, developing innovative disease models to the benefit of our customers.
Please help us find what you looking for, fill out this short massage and we will contact with you shortly.
© all rights reserved to Lahav CRO



